Engineered Structures And Properties
With new strategies, new tools, and new inspirations, researchers are designing ambitious new materials on demand
PEERING INSIDE THE BLACK BOX
Surprisingly little is known about the functioning of many everyday materials. With the help of advanced imaging tools, researchers are beginning to unlock their secrets.

DNA-programmable assembly of 3D-ordered materials from functional nanoparticles. (Image courtesy of Oleg Gang)
Some of the most iconic materials in modern society could have easily never come into existence at all. Saccharin, quinine, safety glass, Super Glue, Teflon—all happy lab accidents, the by-product of inadvertent eureka moments that went on to change the culture.
We at Columbia, with our Data Science Institute and collaborative structure across areas of expertise, have this unique strength in using large data to solve problems.
But such examples are exceptions that prove the rule: materials science is a meticulous art, in which it can take years to refine a single brilliant insight into a working specimen. Researchers have long sought new methodologies to speed the process by systematizing discovery and production techniques.
In recent decades, inverse design—taking a wish list of material properties and working backward to optimal building blocks and precise recipes for connecting them into desired structures—represented a major paradigm shift. Driven by advances in physics and chemistry—and aided by developments in prediction and computation of performance—researchers devised methods to both create sophisticated molecular and nanoscale blocks and build materials with previously unseen properties. But even as engineers gained unprecedented power to achieve desired properties, progress toward rapidly identifying practical pathways for by-design fabrication of materials with targeted functions lagged behind—largely because production techniques remained conceptually similar to the pre-inverse process: moving meticulously from hypothesis to trial and retrial.
Now, materials science is on the verge of another transformative leap that will alter the entire creation cycle. Increased computing power and modeling coupled with breakthroughs in AI and machine learning have dramatically enhanced data analysis of rapid high-throughput experimentation. Combining an evolving ability to exquisitely control material formation across scales with leading-edge multiparameter characterization methods, researchers are vigorously moving toward a day when AI will grant material scientists extraordinary abilities to synthesize desired structures on demand.
“We at Columbia, with our Data Science Institute and broad collaborative structure across areas of expertise in the Engineering School, have this unique strength in using large data to solve problems,” says Sanat Kumar, Bykhovsky Professor of Chemical Engineering. “Designing new materials is one of them.”
Revolutionizing the Process

Simon Billinge with members from his lab, which is developing an ML technique for speeding materials discovery. (Photo by John Abbott)
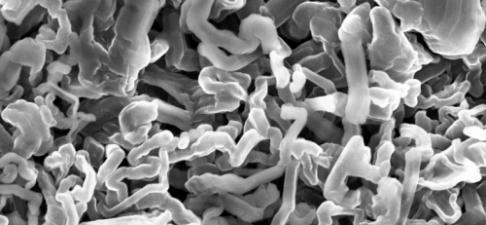
The nanostructure of different components inside a rechargeable AAA battery. Each of the colorful pixels contains a complete mathematical map of the nanostructure. (Figure generated by Marco DiMichiel)
Tackling climate change is a multidisciplinary challenge, but the physical roadblocks to sustainability are often material-related, says professor of materials science and engineering and applied physics and applied mathematics Simon Billinge. Whether the goal is improved fuel cells, better water filtration, or lighter aircraft, the problem starts on the atomic level. In order to manufacture more efficient and durable materials, researchers have been turning to machine learning (ML) as a means for rapidly cycling through millions of potentially viable molecular arrangements. Billinge is devising a new ML methodology that, given a prediction of a desired material, will determine the recipe and parameters to make it.
The trick is to speed up a hit-or-miss process by carrying out and following material syntheses on the fly, while at the same time constantly monitoring the resulting product. Working with chemistry colleagues, both globally and at nearby Stony Brook University, Billinge sets up material synthesis experiments using the intense X-ray beam located inside Brookhaven National Laboratory’s National Synchrotron Light Source II, which can probe the nanostructure of resulting products even as they form, change, and disappear during the process. His group is developing computational infrastructure that will also analyze the data in real time. Those results are then recorded in databases where ML techniques reveal relationships between synthesis parameters and resulting products. The next step will be to “autonomate” that process—meaning that AI programs take over experimental decision-making, dictating strategies for updating synthesis parameters in order to accelerate the pace of discovery.
One of the problems with the current scientific literature is that it is success biased.
The power of these approaches was demonstrated recently when one of Billinge’s students, Chia-Hao Liu, used ML to tease out symmetries in atomic clusters buried in one of the measured signals—the atomic pair distribution function—a feat that previously wasn’t possible.
But in the world of ML, it’s often failed experiments that provide especially valuable data. “One of the problems with the current scientific literature is that it is success biased— those are the only experiments we save for posterity,” says Billinge. To that end, he’s now working with the International Union of Crystallography to create a 21st-century take on the traditional approach to scientific publishing—in essence, a scientific journal that can be read by both humans and machines. Such a digital publication will ultimately house a vast compilation on the synthesis, structure, and characterization of materials— including what doesn’t work. “Researchers around the world can mine all this data to generate predictions,” he says, dramatically speeding materials discovery and helping usher in a sustainable future before the clock runs out on climate change.
Biological Imperatives

From left to right: Sanat Kumar, Kyle Bishop, and Oleg Gang. (Photo by Barbara Alper)

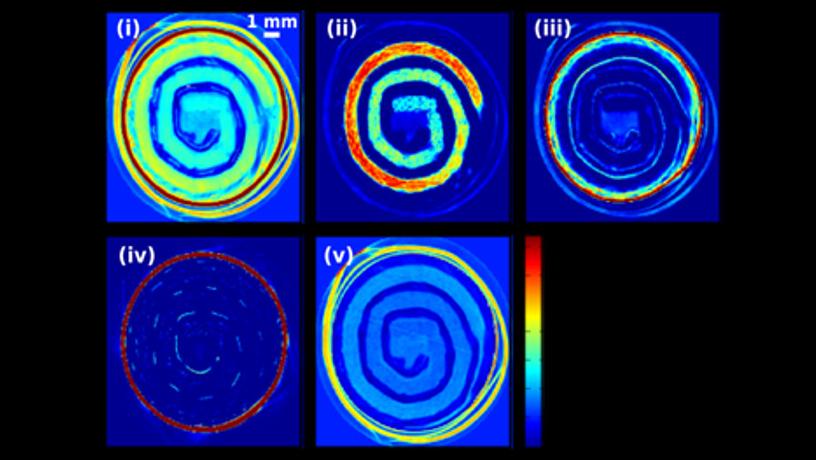
This alignment produces constructive interference in 2D small-angle X-ray scattering, which can be quantitatively correlated with the degree of nanoparticle alignment and the subsequent mechanical reinforcement of the composite plastic. (Image courtesy of Sanat Kumar)
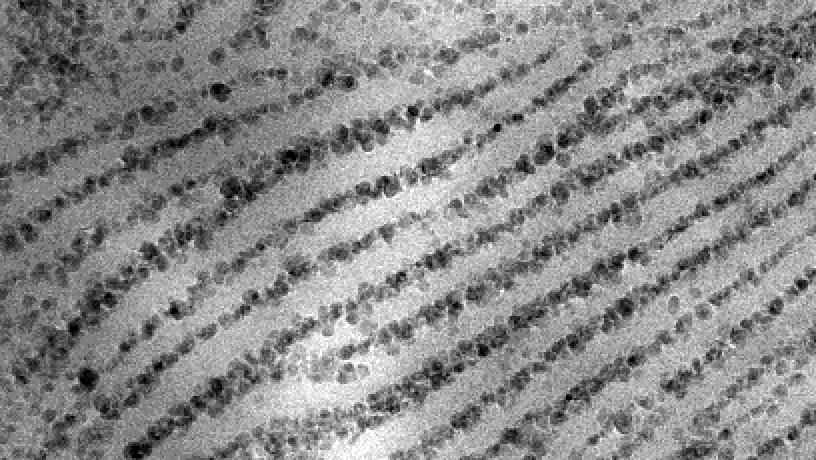
Transmission electron microscopy of isothermally processed polymer nanocomposites inducing nanoparticle assembly into hierarchical regions of the crystal (reminiscent of naturally occurring nacre). (Image courtesy of Sanat Kumar)
We need to discover design principles that are not just conceptually possible, but actually practically feasible.
The ability to observe atomic structure and behavior at the quantum level has provided a tool to copy the way nature solves problems. As Kumar notes, “Biology has a four-billion-year advantage on us. We are using an inverse approach to mimic biological materials and processes, whether our focus is a new class of layered materials, optical fibers, or selective transport membranes.”
Developing an effective, cheap membrane that can be scaled is a personal goal for this chemical engineer. His hometown, Chennai, India, has run out of water, despite that it’s located 14 meters above the Bay of Bengal. His group is developing bio-inspired design rules for hybrid soft materials whose nanostructure combines controllable hydrophilic and hydrophobic characteristics. They are approaching the problem from two angles—one manipulating the nanoarchitecture of lipid cell membranes and correlating outcomes with variables; the other primarily data-centric, establishing rules for an ML platform that will eventually work from a database containing some 20,000 polymer types. An expert in evolutionary algorithms, Kumar formed the Soft Matter Focus Group with Kyle Bishop, an associate professor of chemical engineering, and Oleg Gang, a professor of chemical engineering and applied physics and materials science, in order to bring data science to bear on some of the field’s grandest challenges.
With massive data sets now the norm, “soft materials engineers need a way to navigate an overwhelming set of choices,” says Bishop. “We identified this as a high-level challenge we all share and are creating a framework to leverage for our individual groups and the larger community.”
Together, the group has been developing inverse design tools that leverage automated experiments to mitigate the uncertainty of model predictions. “The analogy is a chess-playing computer program,” Bishop says. “Those programs can make moves beyond the capability of a grand master. We’d love to have the same thing—a machine learning system that can think holistically about a complex material and design maximally informative experiments to guide the design process.”
For Bishop, one immediate application for such a framework comes in the form of colloidal robots. His team researches how external fields can power the autonomous propulsion and navigation of colloidal particles through heterogeneous environments, a line of inquiry that could eventually produce active materials operating inside larger systems in a manner analogous to how the human body repairs itself—something like an immune system for batteries, for instance, where microrobots continuously repair materials from within. The synergy emerging from the Soft Matter Focus Group goes far beyond the group’s framework, however. By combining computational skill, experimental expertise, and the frontier of nanorobotics, their respective labs are cross-pollinating across challenges of energy conversion, materials discovery, and self-assembly.

Liquidhandling robot helps build complex nanomaterials by assisting on assembly of molecularly programmed architectures. (Photo by Barbara Alper)
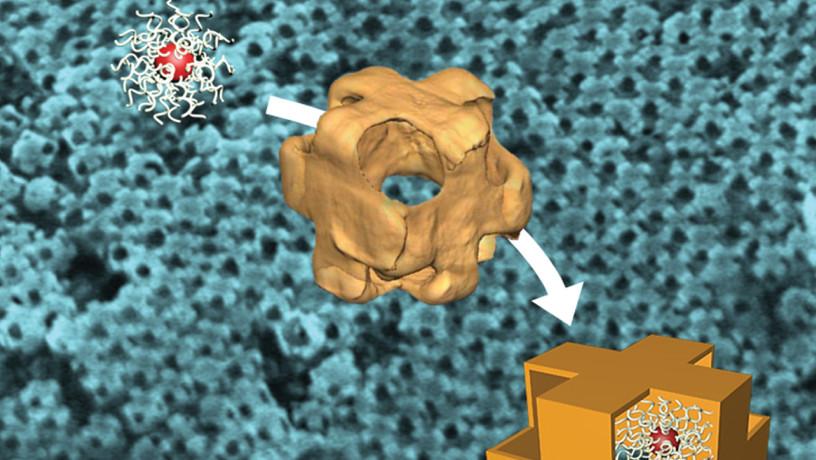
Synthesis and assembly of nanoscale hollow cubes, or so-called “nanowrappers.” Nanowrappers are designed to load and release nanoscale cargo on demand. (Image courtesy of Oleg Gang)
That last element is key to unlocking a second industrial revolution, where materials are no longer created through manual manipulation but instead are programmed to arise spontaneously, based on targeted designs. At the macro level, traditional methods of human-supervised material manufacture are tediously slow, dangerously energy intensive, and often incapable of integrating dissimilar components or controlling structure in space and time. At the nano level, human supervision is well-nigh impossible. “We don’t have tiny tweezers to pick up and organize billions and billions of particles,” Gang says. Some other force has to take the reins. “We need to discover design principles that are not just conceptually possible, but actually practically feasible.”
Recently, Gang’s group revealed a major advance when they developed a DNA platform that demonstrated it’s possible to decouple the assembly process from the material component, using the double helix to stitch together otherwise incompatible nanosubstances such as semiconductors, metals, and proteins into desired architectures. “We wanted to create a material fabrication strategy that’s as versatile as 3D printing, but for the nanoscale,” he says. “And this approach allows us to ‘shape’ and ‘paint’ the desired structure with material ‘colors.’” Such encoded materials would carry instructions not only for how to amalgamate, but also for how to grow and change in structure and function in response to different stimuli, the way living systems do.
“Nature is inspirational for us engineers and scientists,” notes Nanfang Yu.
An associate professor of applied physics with a particular appreciation for nature’s beauty, Yu has long gravitated to the biological world as a source of creativity, both scientifically and artistically. Lately, he’s been translating the insights he’s gleaned there into designing biomimetic coatings and textiles that leverage the unique ability to withstand direct solar heat that have evolved among insects such as the Saharan silver ant, which thrive in desert temperatures averaging around 50 degrees Celsius.
In studying these silver ants, he discovered they are covered along the top and sides of their bodies with hairs shaped like triangular prisms that cool the insects in two ways, via both solar reflection and thermal radiation. These revelations led him to study butterfly wings, on which he’s currently working in collaboration with the Harvard Museum of Comparative Zoology.
About five times thinner than a human hair, butterfly wings are extremely fragile yet remarkably efficient at handling the heat of direct sunlight. Yu discovered that the wings are living organs loaded with mechanical sensors that have a blood supply, which should make them vulnerable to high temperature. Using hyperspectral imaging, he showed that nature-designed micro- and nanoscale structures in special wing scales improve thermal emissivity, keeping the living parts of the wing cooler by 10 degrees to 20 degrees Celsius under full sun than the nonliving membrane that surrounds them.
Based on these observations, Yu has made and tested a radiative-cooling paint that is morphologically different from but physically similar to the thermally efficient nanostructures of butterfly wings. In one application to a roof last summer, an uncoated section of shingles reached 70 degrees Celsius while the coated section never topped 40 degrees. Moreover, a two-story house whose roof was coated consumed 30% less electricity in summer than previously. Yu is extending this cooling strategy to textiles as well and hopes to produce fabric swatches with internally nanostructured heat-efficient fibers within a year.

Yu (left) with student Sajan Shrestha working in front of their optical setup. (Photo by Barbara Alper)

Butterfly specimens being studied in Yu’s lab. (Photo by Barbara Alper)
A Giant Quantum Leap
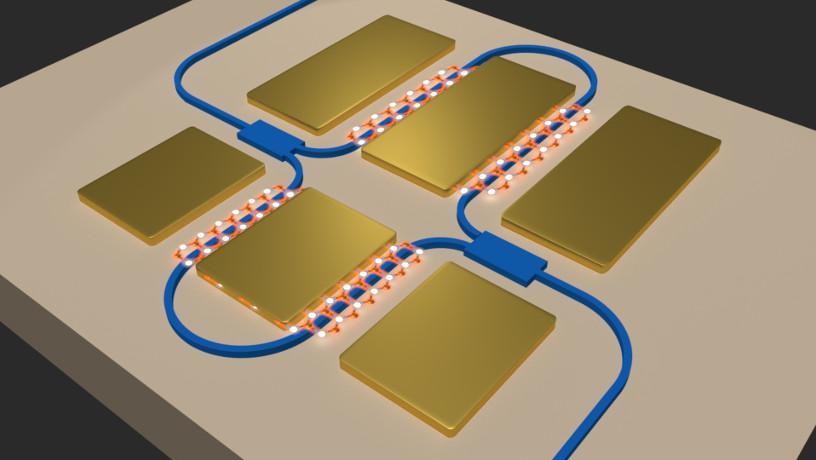
Schematic representation of a photonic circuit, integrated with a one-atom thick TMD to manipulate the speed of light. (Image courtesy of Michal Lipson)
Silicon wafers in Hone’s lab, which are used to create functional devices from new materials. (Photo by John Abbott)
For the past 40 years, researchers have been on an elusive quest to build a viable quantum computer. Before the next decade ends, such machines might well be on their way to commonplace. If that happens, it’ll mean we’ve entered a new era in which applied quantum mechanics doesn’t just revolutionize information security and processing, but also delivers such science-fictional gadgets as sensors that can see through walls or into the future.
“The question is, what is the material base to deliver these beautiful concepts?” says Physics Professor Dmitri Basov.
The answer will be underpinned by new forms of quantum matter that are rendered controllable by gating, magnetic proximity, and nanomechanical manipulation—a process by which researchers take layers just one atom thick and stack them into heterogeneous structures. As principal investigator at Columbia’s Programmable Quantum Materials center (Pro-QM), Basov leads a group bridging the gap between fundamental science and applied physics in exactly this space. Created in 2018 under a $10.7 million Energy Frontier Research Center grant from the Department of Energy, the center ultimately hopes to translate newly discovered laws of physics into desktop devices.
That means that rather than simply identifying such new quantum states, Columbia’s researchers are actively creating them. “Our center participants Cory Dean and James Hone have discovered another tool with very useful capabilities, which is the in operando rotation of these layers,” Basov says. “There’s no analog for that in other material systems.”
Wang Fong-Jen Professor of Mechanical Engineering James Hone serves as both a key Pro-QM researcher and director of Columbia’s related Center for Precision Assembly of Superstratic and Superatomic Solids (PAS3), which is funded by the National Science Foundation. The two centers work together in a strongly complementary fashion; as Pro-QM investigates controlled quantum states, PAS3 creates material platforms enabling such experiments. For instance, PAS3 researchers have not only devised tools capable of twisting graphene into a superconducting state, but also conjured up a means for weaving atomic layers together in a configuration never before seen. Such lines of inquiry open up the possibility of exceptional new tunable properties. “For example, you could separately control how electricity passes through a material versus how heat passes through,” Hone explains.
Many such techniques pioneered at the center make use of another PAS3 specialty: custom-built “superatoms.”
When it comes to desired properties, superatoms—atomic clusters that exhibit single-atom properties—“offer much more design freedom,” Hone notes.
“Say you want something with the reduction potential between lithium and sodium—there’s nothing there on the periodic table,” explains Colin Nuckolls, PAS3 associate director and Columbia’s Higgins Professor of Chemistry. “Those are the two options nature gave you. We’ve demonstrated that there’s an enormous amount of new materials to come out of superatoms.”
PAS3 has become known for its ability to identify and synthesize these clusters, but the group also takes particular advantage of Columbia’s strength in synthesizing molecules. Recently, they built on such work done by center member Latha Venkataraman, Lawrence Gussman Professor of Applied Physics, in the realm of electric-field-driven catalysis. Venkataraman’s team demonstrated that it is possible to convert one molecular isomer into another by applying an electric field. This reaction demonstrated that a field could affect reaction kinetics (speed) and thermodynamics (selectivity)— demonstrating a clear example of “green” chemistry.
At Pro-QM, scientists recently made a discovery in quantum matter research that’s left them breathless. A team led by Michal Lipson, Basov, and Hone has succeeded in isolating a one-atomthick film of transition metal dichalcogenide, which, when voltage was applied while propagating light through it, completely altered the light’s speed.
“This material can slow light drastically or hold it for a very, very long time,” says Lipson, Eugene Higgins Professor of Electrical Engineering and cochair of Pro-QM’s executive committee. “We are now studying this phenomenon so that we can learn to manipulate light and optical properties. It opens the door to many new physics phenomena.”
Because all types of computing—quantum and classical—rely on gates and interconnections, using light to carry information at the chip level has huge advantages. New ways of delaying and manipulating light without losing even one photon would be a giant step forward. “As a community, we have almost all the tools to get closer to actual applications—like quantum computing, and routing transmitting, and routing information—optically,” says Lipson. “The community has been waiting for this.”

James Hone (Photos by John Abbott)

Michal Lipson (Photos by John Abbott)

Colin Nuckolls (courtesy of Colin Nuckolls)

Dmitri Basov (courtesy of Dmitri Basov)